- Point-of-Care
- Central Laboratory
- Life Sciences
- Contract Manufacturing
- Investors
EKF Diagnostics services are built on the core values of flexibility, understanding, and expertise. Because no two jobs are ever the same we offer a tailored approach to every customer, whether you need an end-to-end solution or just require a knowledgeable partner to provide a partial service, we can support your business.
EKF has contract manufacturing sites in Germany and the USA. Across these locations we can provide scalable solutions to support all business types, from start-ups to industry leaders. The solutions include assistance with configuring your product, sourcing materials, providing state-of-the-art formulation and dispensing facilities all the way through to services such as kitting, labelling and packaging.
We understand the importance of quality, we work with our partners to ensure all product and manufacturing requirements are defined and documented to ensure we produce your product to specification whilst maintaining the highest quality standards.
When you partner with EKF you partner with a global diagnostics company that has a proven track record of developing and manufacturing reliable and quality products for a range of markets and industries.
Our Global Manufacturing sites have a range of facilities from controlled Laboratories and White Rooms to ISO Class 7 Clean rooms. EKF will ensure your product requirements are understood allowing the appropriate manufacturing environment to be identified.
Assess product feasibility and produce small scale production batches in EKF’s Pilot Production Lab. Whether you want to formulate a new reagent or pack a new kit, our skilled scientists and operatives will work with you to develop fit for purpose production and test protocols to take your concept to the next stage.
EKF Diagnostics can offer scalable contract formulation. All of our sites have the capability to formulate from 20 to 800 Litre mixes with experience in formulation of many reagent types from Phosphate Buffered Saline to complex reagents for molecular kits.
Our verified dispensing lines are controlled by in-process checks to ensure accurate filling. Our semi-automated pumps allow a variety of volumes of reagents to be dispensed into a range of vessels. With options for manual dispensing lines for process flexibility or automated filling for speed, EKF will have the dispensing solution that best suits your project.
Our adaptable white room suites can be used for manual or automated packing to maintain a fast and flexible workflow.
If your product is required to be manufactured within a clean room environment our sites offer ISO Class 7 controlled clean rooms as well as a fully trained team of operatives.

Contract dispensing suites

ISO Class 7 clean room
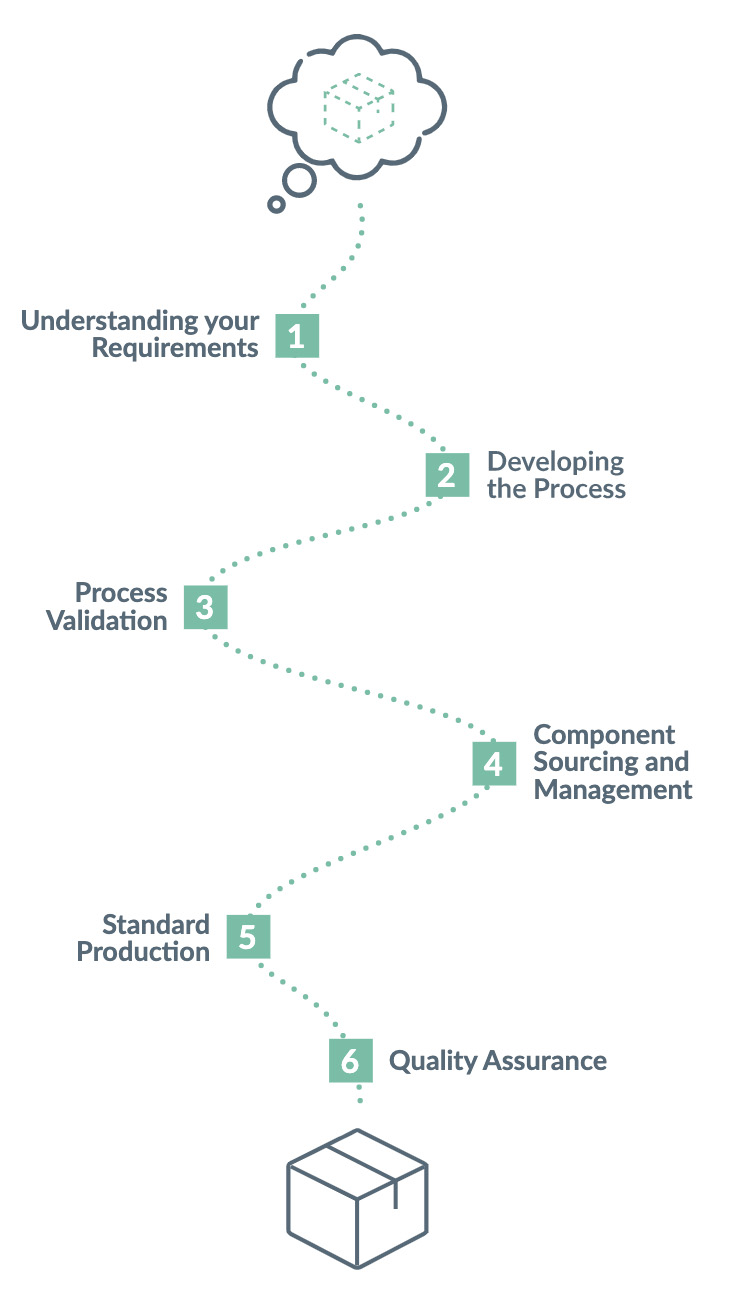
Steps to outsourcing your manufacturing:
EKF will become a key part of your team, we aim to understand the intricacies of your product enabling us to determine the best build solutions for your business.
We understand that your product is continuously developing and improving, our product specification is a live document that we use to ensure your ideas are captured and controlled throughout this process whilst guaranteeing clear communication between our team and yours.
On completion of step 2, we will have a clear idea on the necessary In-process and Final checks to ensure high quality product is consistently produced.
EKF encourage the production of 3 validation batches to assess the production process, this enables us to demonstrate that our production process produces product aligned with your requirements.
Simply send us a Purchase order stating the product code and required quantity and we will manage the rest.
Our manufacturing facilities are fully equipped for production and have skilled operators trained in Lean Manufacture techniques.
Be kept up to date on your order status with our transparent and responsive Customer Service Team.
EKF’s global reach and industry presence provides access to a network of suppliers to source high quality raw materials at the best prices.
EKF can design and develop custom label solutions for your product and control and maintain label databases during product manufacture.
Utilise our experienced Quality and Regulatory teams to understand the regulatory pathway for your product.
Work alongside our experienced Product Managers to develop your idea and understand the market and commercialisation requirements.
Our in-house graphic design team can bring your product to life with packaging design based around your brand identity. We can also help with the more mundane tasks like designing IFUs and other documentation.
We have a global network of specialist IVD distributors who can be accessed to support the commercialisation of your product around the world, subject to product registration.

"EKF Diagnostics have been instrumental in our development as a company. They worked above and beyond their remit to ensure the successful completion of our pivotal milestones. They worked closely with our team to provide solutions and guidance navigating the tricky start-up landscape, whilst providing top-tier products.
In the recent months, they have helped us produce an RUO product for our first client, working to very time-sensitive deadlines on short notice to ensure we made it to the finish line. Furthermore, they have been extremely receptive and adaptive to our evolving product, helping us every step of the way. This continual support is going to be extremely useful in achieving CE validation in the coming month, and we have no doubt that EKF are going prove to be influential in us achieving this."
Elliot Quigley, Project Manager,
Proton Dx

"EKF Diagnostics’ contract manufacturing has been a key part of the exponential growth of Longhorn's core product, PrimeStore MTM, since the start of the SARS-CoV-2 pandemic. Their knowledgeable team of chemists rapidly scaled manufacturing lot sizes from 200 to 1600 liters. Their executive team devoted significant resources to further expand capacity and add additional manufacturing sites to support our growth. As volumes subsequently ebbed and flowed, EKF was supportive of our needs and production scheduling. Sourcing from and working with EKF allowed us to devote our capital to other parts of the business while ensuring that we never ran short of product for our global customers."
Jeff Fisher, President
Longhorn Vaccines and Diagnostics LLC
Copyright © 2024 | Powered by Intergage

Please note: Not all products are available in all countries. Please check availability.
Newsletter
Keep up to date with all our activities, events, exhibitions, promotions, investor news & more.